Danopharm Chemicals Walk in Interview 2026 | Ankleshwar
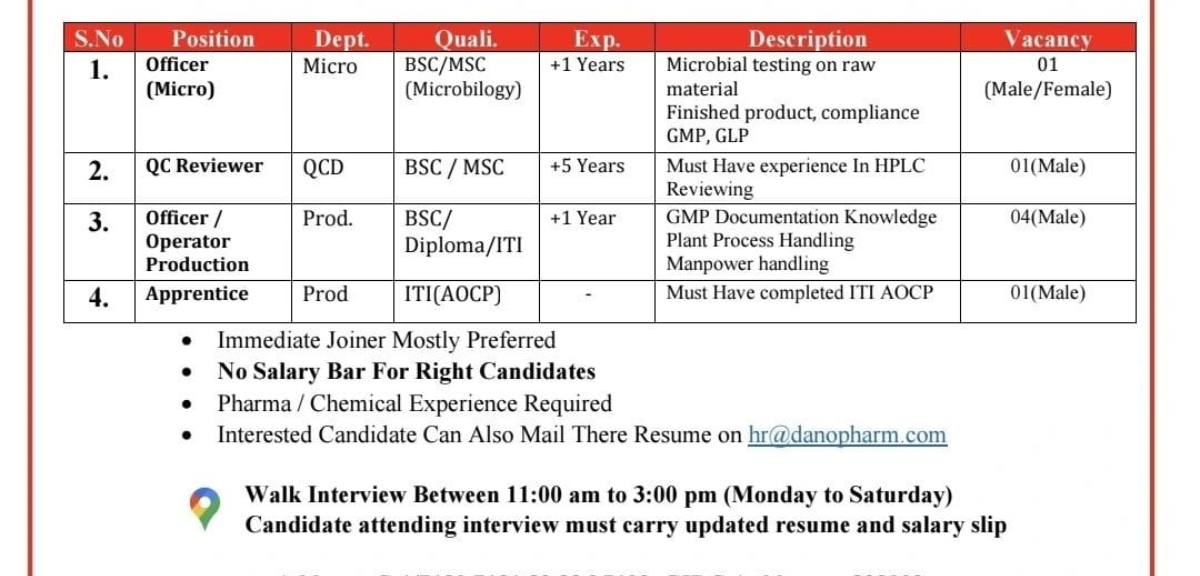
WhatsApp Group Join Now Instagram Group Join Now Danopharm Chemicals Walk in Interview 2026 notification has been announced for multiple pharma positions at their manufacturing facility in Ankleshwar, Gujarat. Candidates with experience in Microbiology, Quality Control, Production, and ITI trades are invited to attend the walk-in interview. This is an excellent opportunity for pharma and chemical industry professionals seeking stable career growth in a GMP-compliant organization. About Danopharm Chemicals Pvt. Ltd. Danopharm Chemicals Pvt. Ltd. is an Indian manufacturer of pharmaceutical bulk drugs and intermediates. Established in 1995, the company operates a modern manufacturing facility in Ankleshwar, Gujarat, equipped for complex chemical reactions and operating under Good Manufacturing Practices (GMP). The company serves global markets and also provides contract manufacturing services. Open Positions Officer (Micro) QC Reviewer Officer / Operator – Production Apprentice Additional Hiring Highlights Micro QC Production Walk in interview 2026 Details Interview Timing:11:00 AM to 3:00 PM (Monday to Saturday) Documents Required: Interview Address C-1/7130, 7131, 32, 33 & 7102GIDC Ankleshwar – 393002District Bharuch, Gujarat