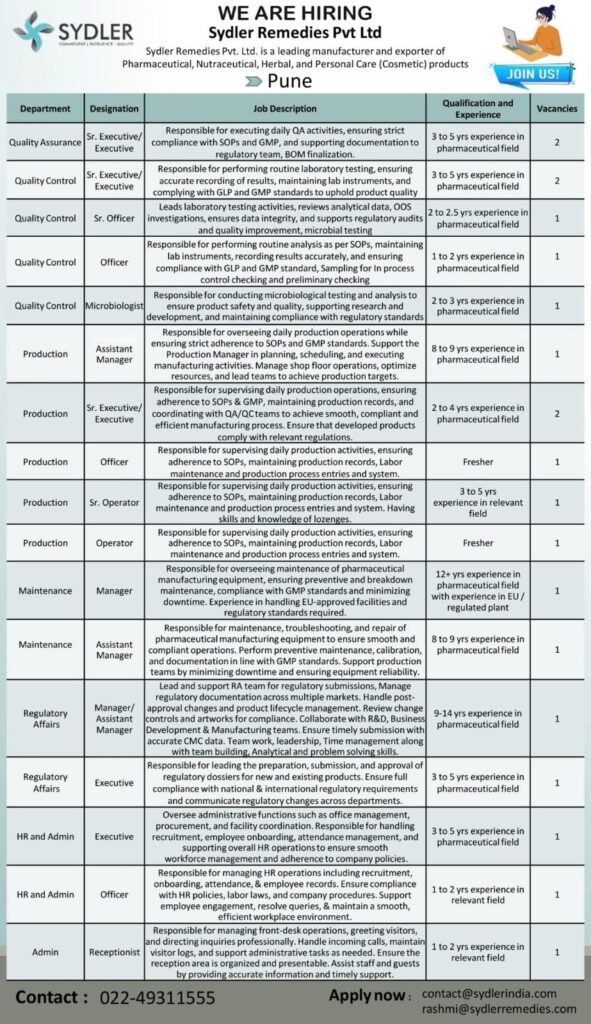
QA QC Production RA Jobs in Pune
Looking for exciting career opportunities in the pharmaceutical sector? Sydler Remedies Pvt Ltd is hiring for multiple departments including Quality Assurance, Quality Control, Production, Maintenance, Regulatory Affairs, and HR/Admin at its facility in Pune.
This is a great opportunity for both freshers and experienced professionals to join a fast-growing pharma company.
Job Overview
- Company: Sydler Remedies Pvt Ltd
- Location: Pune, Maharashtra
- Departments: QA, QC, Production, Maintenance, RA, HR/Admin
- Experience Required: Fresher to 14+ Years
- Job Type: Full-time Opportunities
Department-wise Job Openings
Quality Assurance (QA)
- Designation: Sr. Executive / Executive
- Experience: 3 to 5 years
- Vacancies: 2
- Key Responsibilities:
- QA activities, SOP & GMP compliance
- Documentation and regulatory support
- BOM finalization
Quality Control (QC)
- Sr. Executive / Executive
- Experience: 3–5 years | Vacancies: 2
- Sr. Officer
- Experience: 2–2.5 years | Vacancies: 1
- Officer
- Experience: 1–2 years | Vacancies: 1
- Microbiologist
- Experience: 2–3 years | Vacancies: 1
Skills Required:
- GLP & GMP compliance
- Analytical testing (HPLC, lab instruments)
- Microbial testing & data integrity
Production Department
- Assistant Manager
- Experience: 8–9 years | Vacancies: 1
- Sr. Executive / Executive
- Experience: 2–4 years | Vacancies: 2
- Officer
- Experience: Fresher | Vacancies: 1
- Sr. Operator
- Experience: 3–5 years | Vacancies: 1
- Operator
- Experience: Fresher | Vacancies: 1
👉 Responsibilities:
- SOP & GMP compliance
- Production planning & execution
- Equipment handling and process monitoring
Maintenance Department
- Manager
- Experience: 12+ years (EU regulated plant preferred) | Vacancies: 1
- Assistant Manager
- Experience: 8–9 years | Vacancies: 1
👉 Skills:
- Preventive & breakdown maintenance
- Equipment calibration
- GMP compliance
Regulatory Affairs (RA)
- Manager / Assistant Manager
- Experience: 9–14 years | Vacancies: 1
- Executive
- Experience: 3–5 years | Vacancies: 1
👉 Responsibilities:
- Regulatory submissions
- Lifecycle management
- Compliance with global regulations
HR & Admin
- Executive
- Experience: 3–5 years | Vacancies: 1
- Officer
- Experience: 1–2 years | Vacancies: 1
Admin Department
- Receptionist
- Experience: 1–2 years | Vacancies: 1
How to Apply?
Send your updated resume to:
Contact: 022-49311555